Thermos Index Thermodynamics Heat Engines
|
Introduction The function of a heat engine is to transform energy which is supplied by heat transfer
into mechanical energy and make it available for doing useful work...Any system which enables
a substance or working agent to undergo a cyclic process by receiving and rejecting energy by
heat transfer with some of this energy converted into work is called a heat engine.
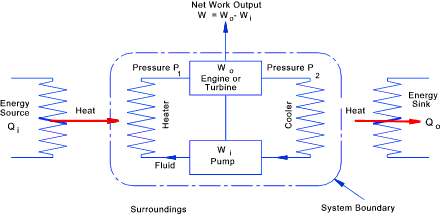
For the typical heat engine, as shown, the working agent is assumed to complete a cyclic process. There is no net gain or loss of energy within the boundary (Δ E = 0). This is in accordance with the First Law of Thermodynamics or the law of conservation of energy. Qi + Wi = Qo + Wo Also the net work done by the engine is equal to (the work out - the work expended ). W = Wo - Wi.....= Qi - Qo The efficiency of the heat engine is the ratio of the useful work output and the energy input i.e.
All heat engines have the following essential components
All other parts of the heat engine are provided for the successful operation and functioning
of the three essential components identified above. |
Thermodynamic /Heat Transfer Links
|
|
Thermos Index